FACTS ABOUT BENZENE
Benzene is a molecule with the chemical formula of C6H6, and because of this it has a molar mass of 78 grams/mole. Benzene is most often seen as a liquid with no color, and has a scent very similar to that of gasoline. This molecule has other interesting qualities such as is solubility in water and its melting point at the temperature of 5.33 degrees Celsius. Benzene has a boiling point at a degree of 176.2 degrees Fahrenheit.
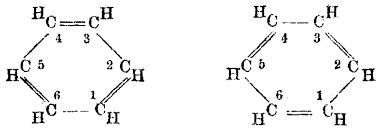
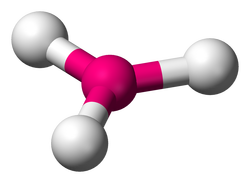
Benzene has a six carbon ring structure which actually makes it a very stable element due to the equal lengths of all the hydrogen and carbon bonds. Benzene is also soluble in water and Benzene itself has a molecular shape of Trigonal planar, which means that the molecule exists in a triangular shape across a single plane in space, much like the figure below.
Benzene is a molecule with the chemical formula of C6H6, and because of this it has a molar mass of 78 grams/mole. Benzene is most often seen as a liquid with no color, and has a scent very similar to that of gasoline. This molecule has other interesting qualities such as is solubility in water and its melting point at the temperature of 5.33 degrees Celsius. Benzene has a boiling point at a degree of 176.2 degrees Fahrenheit.
Benzene has a six carbon ring structure which actually makes it a very stable element due to the equal lengths of all the hydrogen and carbon bonds. Benzene is also soluble in water and Benzene itself has a molecular shape of Trigonal planar, which means that the molecule exists in a triangular shape across a single plane in space, much like the figure below.
Benzene itself is formed during natural occurrences such as fires and volcanoes, but most of the Benzene that people come in contact with is actually from human activities. Unexpectedly, the chemical of Benzene can be disposed of in many ways. If in small quantities and/or liquid, one could absorb the Benzene into sand or dirt, then dispose of it in a clean and conscious land fill. Also, one could absorb the Benzene into a pile of dirt or sand, and then cautiously ignite it so that it burns away (in the case of liquid). But in larger amounts, you would actually have to atomize it in a combustion chamber. Atomizing is the process by which one converts or reduces something into very tiny particles or units. By doing so, it is making Benzene much less potent and suitable for disposal in large quantities.
