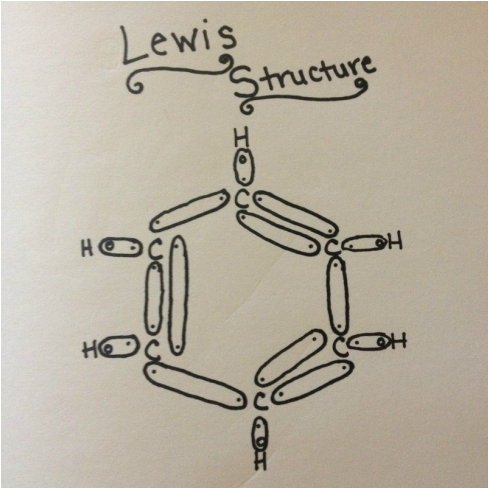
The Lewis Structure of Benzene is shown here. As stated before, Benzene is a carbon based molecule, made up of six carbon atoms, along with six hydrogen atoms. They bond together in a such a way that the carbons form a ring, some with double bonds, and with each carbon there is a hydrogen filling their last available bonding space. The AX structure of Benzene is not found on the VSEPR table because it is a closed ring of atoms, therefore it is called a cydohexane molecule.
As far as inter molecular forces which Benzene would possess, it would all depend on the molecule with which it was having contact with. No matter what the other molecule was, Benzene and said molecule would have the force of dispersion, which acts on all molecules. And then from this, one would assume that depending on the composition of the other molecule, Benzene and it would have the ability to Hydrogen Bond. Dipole-dipole would not occur because Benzene is not a polar molecule.
As far as inter molecular forces which Benzene would possess, it would all depend on the molecule with which it was having contact with. No matter what the other molecule was, Benzene and said molecule would have the force of dispersion, which acts on all molecules. And then from this, one would assume that depending on the composition of the other molecule, Benzene and it would have the ability to Hydrogen Bond. Dipole-dipole would not occur because Benzene is not a polar molecule.